
PCOS is one of the most common endocrine disorders, affecting up to 20% of women of reproductive age. The diagnostic criteria for PCOS include chronic oligomenorrhea or anovulation, hyperandrogenism, and polycystic ovarian morphology. PCOS is associated with an increased risk of developing hypertension, dyslipidemia, type 2 diabetes, and heart disease. Insulin resistance is another common feature of PCOS in both overweight and lean women, and it is often treated with insulin sensitizers like metformin. Over the last decade, myo-inositol, an isomerized and dephosphorylated precursor of glucose-6-phosphate, has been used more and more as a natural insulin sensitizer. The role of myo-inositol in the pathophysiology of PCOS has been the focus of recent studies. It has been suggested that insulin resistance in PCOS women can be attributed to a deficiency of myo-inositol’s intracellular metabolites, D-chiro-inositol (DCI) and inositol-phosphoglycan (IPG), mediators of insulin action. It has also been postulated that patients with PCOS have a higher urinary clearance of DCI. In addition to its role in insulin sensitivity, DCI functions as an intracellular messenger in mammalian oocytes, playing a role in the follicular milieu, meiotic resumption, and oocyte maturation.1
Due to the key role of insulin in the syndrome etiopathology, for many years, insulin sensitizers such as metformin, pioglitazone, or troglitazone have been considered as possible therapeutic options in the management of these problems. Metformin has been used in the last time on patients with a hyperinsulinemic status for the improvement of ovarian dysfunction with consecutive anovulation, irregular menstrual cycles, and infertility problems. Nevertheless metformin, when used in the therapeutic dose range, was shown to have several side effects such as flatulence, diarrhea, and nausea, so that many patients are unable to use this treatment option in gynecology for a longer period of time.2
Therefore, in parallel to the common use of metformin and other insulin sensitizer agents for the treatment of PCOS, in the recent years, other therapeutic alternatives have been investigated.
Treating women with myo-inositol or DCI has been shown to reduce androgen levels, help restore ovulatory function, lower blood pressure, and decrease triglyceride levels. This study confirms prior studies that have shown that myo-inositol or DCI administration improves endocrine parameters and insulin sensitivity, more so in women with higher fasting insulin levels, defined by a fasting insulin level greater than 12 µU/mL. Although weight loss and dietary interventions are also known to improve insulin sensitivity, hyperandrogenism and restore ovulation in women with PCOS, studies investigating the potentially additive benefit of combining lifestyle interventions with myo-inositol would be beneficial. Myo-inositol is a safe and effective natural medicine for improving insulin resistance and it should be recommended in conjunction with other positive lifestyle modifications for the management of PCOS and insulin resistance.3
Myoinositol in PCOS
DCI Paradox:
Confirmed by a recent study showing that the oocyte quality and ovarian response have been progressively worsened by the increased dosage of D chiro inositol(DCI).
Therefore, DCI supplementation is not recommendable for several reasons:
(a) High doses of DCI/day have been considered toxic to ovaries and oocyte maturation
(b) DCI is not converted into MI; therefore, the specific action exerted by the last one would be Lost
Since MI competes with glucose for cellular transporters, a low MI intracellular uptake and increased urinary loss of MI may lead to high blood glucose levels. It can be supposed that adequate MI intake and body storages might derive the epimerase by substrate mass action inducing DCI synthesis. This agrees with the clinical evidence that reported the beneficial effect exerted by MI and DCI combined at 40:1 ratio in PCOS patients.5
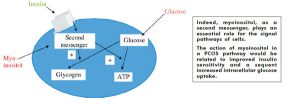
It is noteworthy that improvement of IR and reduction of circulating insulin are key therapeutic targets in PCOS, enhancing fertility and reducing the lifelong risk for type 2 diabetes and early cardiovascular disease. Besides metformin, MI is now considered as a further insulin-sensitizing supplement which could benefit women with PCOS. Exercises and a healthy nutritional lifestyle have widely been gathering attention in the management of PCOS.
Though DCI is crucial for glycogen synthesis; MI has an action at ovarian level. Restoring MI levels will improve ovarian response to FSH; since MI is responsible for glucose cell intake, energy status will be restored as well, it is able to improve oocyte quality.6
Evidence based studies
After the 8-week myo-inositol treatment, there was a significant reduction in LH/FSH ratio, FSH, prolactin, androstenedione, testosterone, insulin, and BMI compared to baseline; there was also a significant increase in the glucose/insulin ratio (8.4±0.9 to 12.1±0.2, P<0.01). In consideration of baseline fasting insulin levels, myo-inositol treatment induced similar changes in both groups but only patients in group B demonstrated significant reduction in fasting insulin levels (20.3±1.8 to 12.9±1.8 µU/mL, P<0.00001). No side effects or adverse events were observed in any of the study participants; however, a prior study demonstrated that when myo-inositol was dosed at 12 grams per day, patients experienced gastrointestinal distress like nausea, diarrhea, and flatulence.7
Despite the clear limitations of the observational study, there are reliable available data, since a wide range of patients can be analyzed. This study could show that a new treatment option for patients with a PCOS and infertility is available. Seventy % of the patients restored ovulation after the treatment. Furthermore, the achieved pregnancy rates are at least in a range equivalent to or even superior to those reported by the use of the insulin sensitizer metformin. Karimzadeh and Javedani described a pregnancy rate of 14.4% in a cohort of 90 women and Legro et al. Of 12.3% in a cohort of 75 women with PCOS.8
The interesting results that the study has shown seem to be related to the mechanism of action of myoinositol. The administration of this molecule, acting as a direct messenger of insulin signaling and improving the glucose tissues uptake, could improve the insulin resistance status of PCOS women, restoring indeed their hormonal status and restoring the ovulation process.9
Important evidence is also related to the difference of myoinositol and metformin in terms of safety profile and compliance for patients. In patients under metformin, side effects have been commonly reported, in particular from mild up to severe gastrointestinal side effects, such as abdominal pain, nausea, and diarrhea. Only in rare cases, very severe side effects as lactic acidosis have been found. On the other side, myoinositol seems to be a safe and well-tolerated approach, anyhow able to give similar results of metformin in terms of clinical efficacy.10
In fact, many studies have demonstrated in the last months that an improvement in the rates of ovulation and regularization of menstrual cycles was obtained by the combined use of 4 g myoinositol with 400 μg folic acid per day. Gerli et al. could show in a prospective study that the group of patients receiving myoinositol + folic acid experienced in 82% of the cases an ovulation, whereas this was only observed in 63% of the cases in the group of patients which received a placebo. By the same way 70% of the patients of the myoinositol group developed regular menstrual cycles after 16 weeks of treatment, whereas only 13% of the women did it in the placebo group.11, 12
In a study of Raffone et al., where a comparison between the administration of myoinositol (2 × 2000 g + Folic Acid 200 μg per day) and the administration of metformin (1500 mg per day) in women with a PCO syndrome was performed, it could be shown that the number of pregnancies was clearly higher in the myoinositol group than in the metformin group of patients.
Some other studies upon others have shown the efficacy of myoinositol in the improvement of the fertility of PCOS patients due to its improvement of the insulin resistance of these women.
Many studies have been performed that show that the treatment with myoinositol + folic acid in the classical dosage (2 × 2000 g myoinositol + 200 μg folic acid per day) leads to significant positive changes of metabolic and hormonal parameters. Costantino et al. could show in a double-blinded, placebo controlled study that myoinositol led to a statistically significant improvement of the blood pressure, triglycerides, cholesterol, glucose, and insulin values after a 75 mg oral glucose tolerance test. These improvements were achieved after a treatment period of 16 weeks. The evaluated hormonal values showed a significant decrease of the total and free testosterone serum levels and at the same time the progesterone levels, as a marker of ovulation, experienced a significant rise in the group that received myoinositol . This could show that myoinositol did lead not only to positive changes in metabolic parameters but also to a reduction of elevated androgenic values and subsequently to an improvement of skin problems such as acne or hirsutism.
References
- Genazzani A. D., Lanzoni C., Ricchieri F., Jasonni V. M. Myo-inositol administration positively affects hyperinsulinemia and hormonal parameters in overweight patients with polycystic ovary syndrome. Gynecological Endocrinology. 2008;24(3):139–144. doi: 10.1080/09513590801893232. [PubMed][Cross Ref]
- Croze M. L., Soulage C. O. Potential role and therapeutic interests of myo-inositol in metabolic diseases. Biochimie. 2013;95(10):1811–1827. doi: 10.1016/j.biochi.2013.05.011. [PubMed][Cross Ref]
- Baillargeon J.-P., Iuorno M. J., Nestler J. E. Insulin sensitizers for polycystic ovary syndrome. Clinical Obstetrics and Gynecology. 2003;46(2):325–340. doi: 10.1097/00003081-200306000-00011. [PubMed][Cross Ref]
- Genazzani A. D., Battaglia C., Malavasi B., Strucchi C., Tortolani F., Gamba O. Metformin administration modulates and restores luteinizing hormone spontaneous episodic secretion and ovarian function in nonobese patients with polycystic ovary syndrome. Fertility and Sterility. 2004;81(1):114–119. doi: 10.1016/j.fertnstert.2003.05.020. [PubMed][Cross Ref]
- Strugaru A. M., Botnariu G., Agoroaei L., grigoriu I. C., Butnaru E. Metformin induced lactic acidosis—particularities and course. Revista Medico-Chirurgicala a Societatii de Medici si Naturalisti din Iasi. 2013;117(4):1035–1042. [PubMed]
- Kim C.-H., Han K.-A., Oh H.-J., et al. Safety, tolerability, and efficacy of metformin extended-release oral antidiabetic therapy in patients with type 2 diabetes: an observational trial in Asia. Journal of Diabetes. 2012;4(4):395–406. doi: 10.1111/j.1753-0407.2012.00220.x. [PMC free article][PubMed] [Cross Ref]
- Clements R. S., Jr., Darnell B. Myo-inositol content of common foods: development of a high-myo-inositol diet. The American Journal of Clinical Nutrition. 1980;33(9):1954–1967. [PubMed]
- Reynolds J. E. F. Martindale: Martindale-the Extra Pharmacopoeia.30th. Pennsylvania, Pa, USA: Rittenhouse Book Distributors; 1993. (An Isomer of Glucose that Has Traditionally Been Considered to Be a B Vitamin Although it Has an Uncertain Status as a Vitamin and a Deficiency Syndrome Has not Been Identified in Man).
- Baillargeon J.-P., Nestler J. E., Ostlund R. E., Apridonidze T., Diamanti-Kandarakis E. Greek hyperinsulinemic women, with or without polycystic ovary syndrome, display altered inositols metabolism. Human Reproduction. 2008;23(6):1439–1446. doi: 10.1093/humrep/den097. [PMC free article][PubMed] [Cross Ref]
- Hooper N. M. Glycosyl-phosphatidylinositol anchored membrane enzymes. Clinica Chimica Acta. 1997;266(1):3–12. doi: 10.1016/S0009-8981(97)00161-7. [PubMed][Cross Ref]
- Carlomagno G., Unfer V. Inositol safety: clinical evidences. European Review for Medical and Pharmacological Sciences. 2011;15(8):931–936. [PubMed]
- Karimzadeh M. A., Javedani M. An assessment of lifestyle modification versus medical treatment with clomiphene citrate, metformin, and clomiphene citrate-metformin in patients with polycystic ovary syndrome. Fertility and Sterility. 2010;94(1):216–220. doi: 10.1016/j.fertnstert.2009.02.078. [PubMed][Cross Ref]

